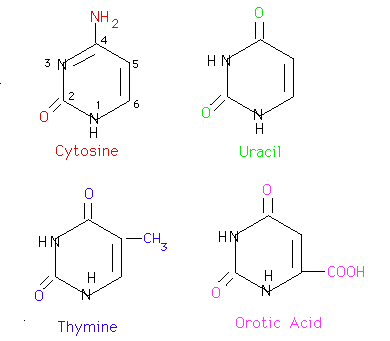
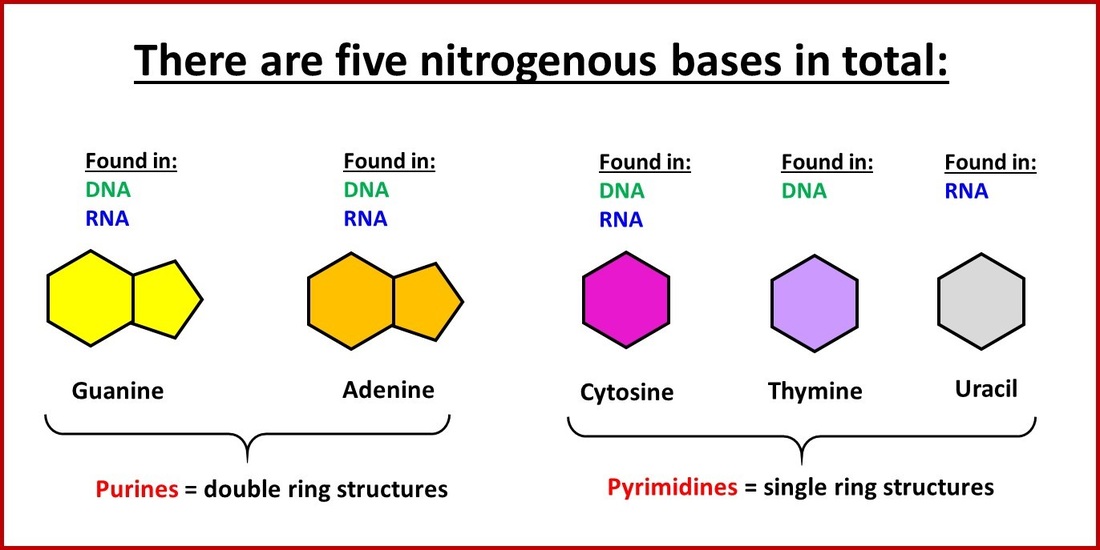
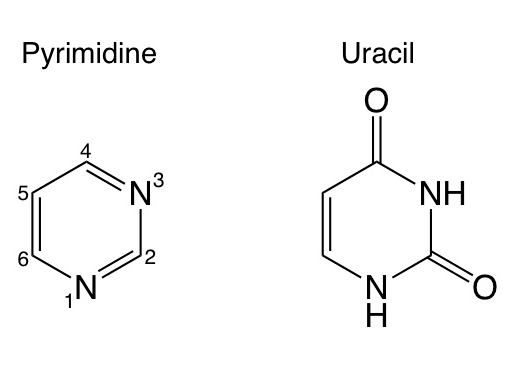
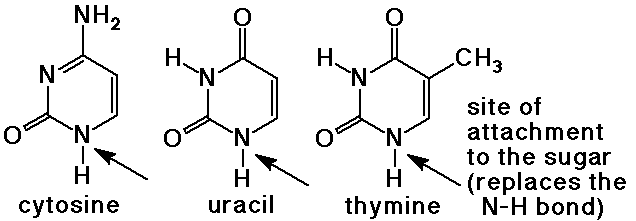
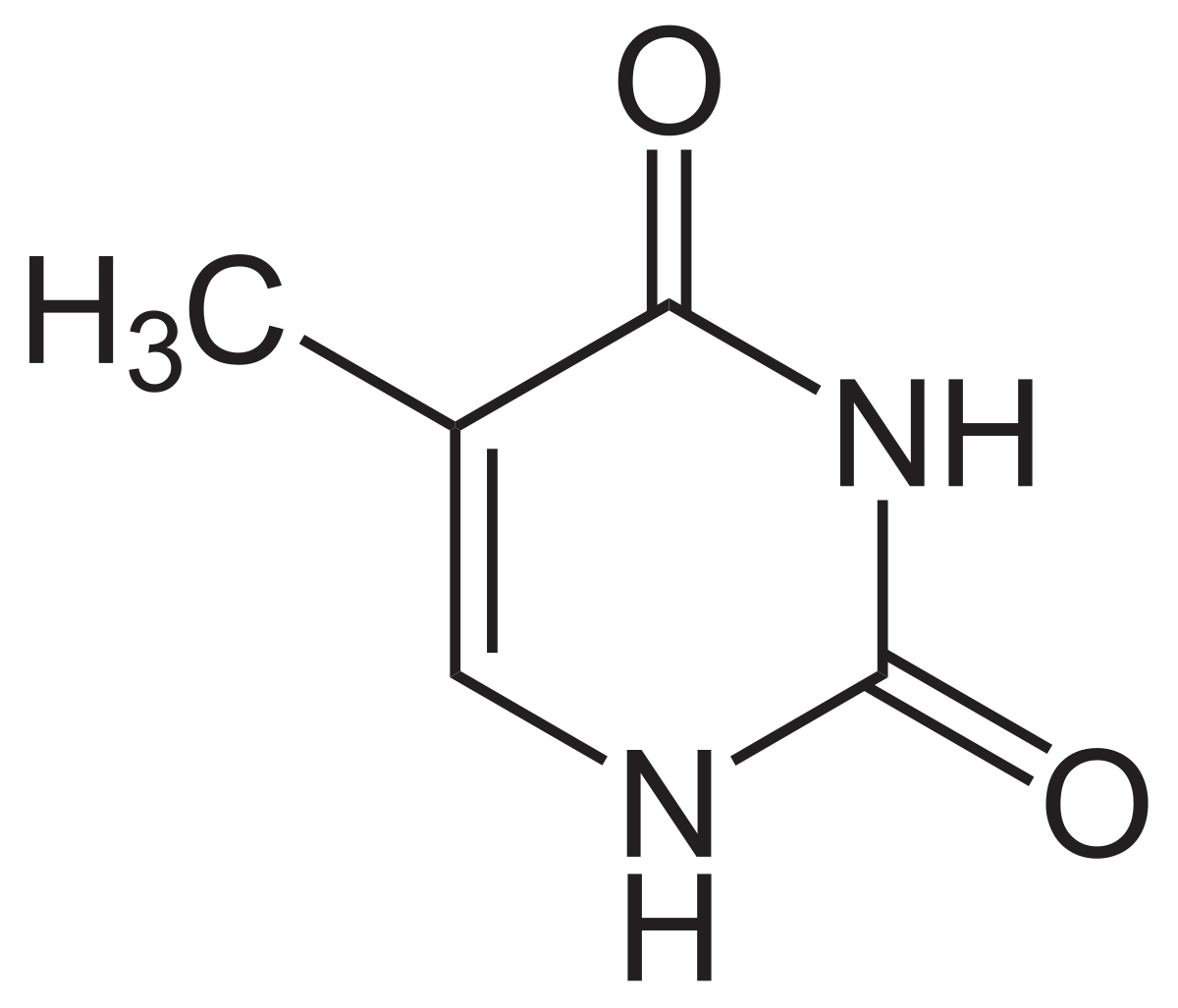
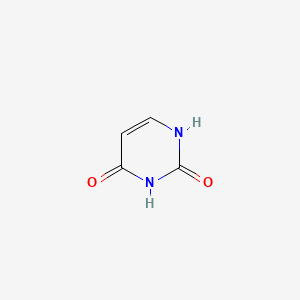
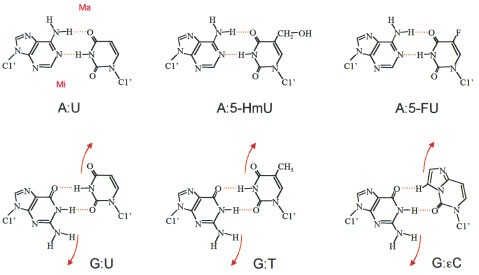
Solvation Structure of Uracil in Ionic Liquids - Norman - 2016 - ChemPhysChem - Wiley Online Library

Table 3 from Synthesis of Uracil-Iodonium(III) Salts for Practical Utilization as Nucleobase Synthetic Modules | Semantic Scholar
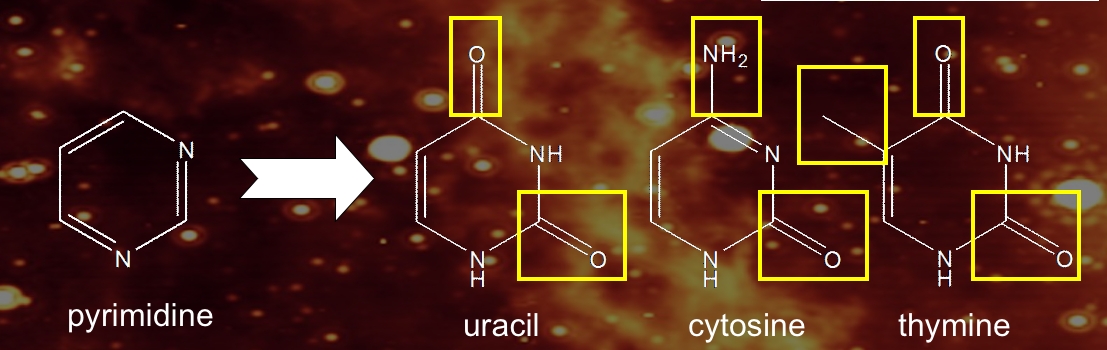
The Astrophysics & Astrochemistry Laboratory: Nucleobases and Their Production during the Photolysis of Astrophysically Relevant Ices

Uridine Is A Glycosylated Pyrimidine Analog Containing Uracil Attached To A Ribose Ring Via A N1glycosidic Bond Stock Illustration - Download Image Now - iStock

Uridine Is A Glycosylated Pyrimidine Analog Containing Uracil Attached To A Ribose Ring Via A N1glycosidic Bond Stock Illustration - Download Image Now - iStock
Effect of reduction of the tunicamycin uracil ring on binding to the... | Download Scientific Diagram
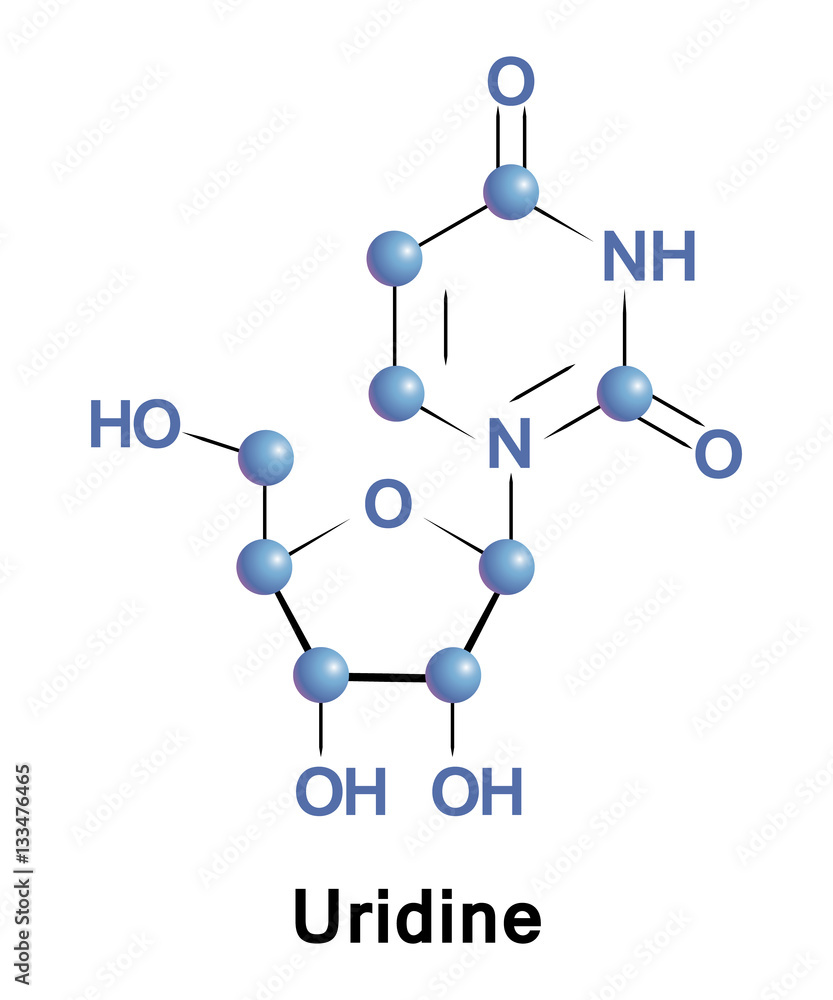
Uridine is a glycosylated pyrimidine-analog containing uracil attached to a ribose ring via N1-glycosidic bond. It is one of the five standard nucleosides which make up nucleic acids Stock Vector | Adobe

A Plausible Mechanism of Uracil Photohydration Involves an Unusual Intermediate | The Journal of Physical Chemistry Letters

Uracil ring opening in the reaction of 5-formyl-2′-deoxyuridine with primary alkyl amines - ScienceDirect

Figure 1 from Nitrosative cytosine deamination. An exploration of the chemistry emanating from deamination with pyrimidine ring-opening. | Semantic Scholar